





Disc herniation is a painful condition caused by the protrusion of the intervertebral disc which compresses the surrounding nerve roots causing "radiculopathy". Anatomically, the intervertebral disc is composed of a pulpy nucleus of gelatinous consistency and of an anulus fibrosus which has the structural function of containing the nucleus. When the anulus is damaged, the nucleus emerges from its seat, protruding towards the vertebral canal.
Traumatic events or, in most cases, the degeneration of the anatomical structure of the disc linked to ageing phenomena are the main causes of disc herniation.
The main symptom is the pain which can be perceived at the level of the area actually affected by the hernia, but which can also radiate to the entire nervous path of the nerves involved. Disc herniation is more frequent at the lumbar and cervical level - lumbar hernias are 15 times more frequent than cervical ones - these two types of hernias differ also in the location of the pain: affecting the neck and arms for cervical hernias, while lumbar disc protrusions cause irritation of the sciatic nerve and therefore sciatica. The level of suffering is generally progressive and can determine important limitations such as the inability to perform physical activity, which in turn causes a possible acceleration of further degenerative events which lead, for example, to an increase in pain and body weight.
The diagnosis involves the physical examination of the patient, evaluating the site of the pain, the state of the reflexes, the sensitivity, strength and muscle trophism. X-ray examinations such as magnetic resonance imaging, which also allows the assessment of the involvement of the surrounding nerve structures and the degree of inflammation of the segment, are functional for confirming the disease.
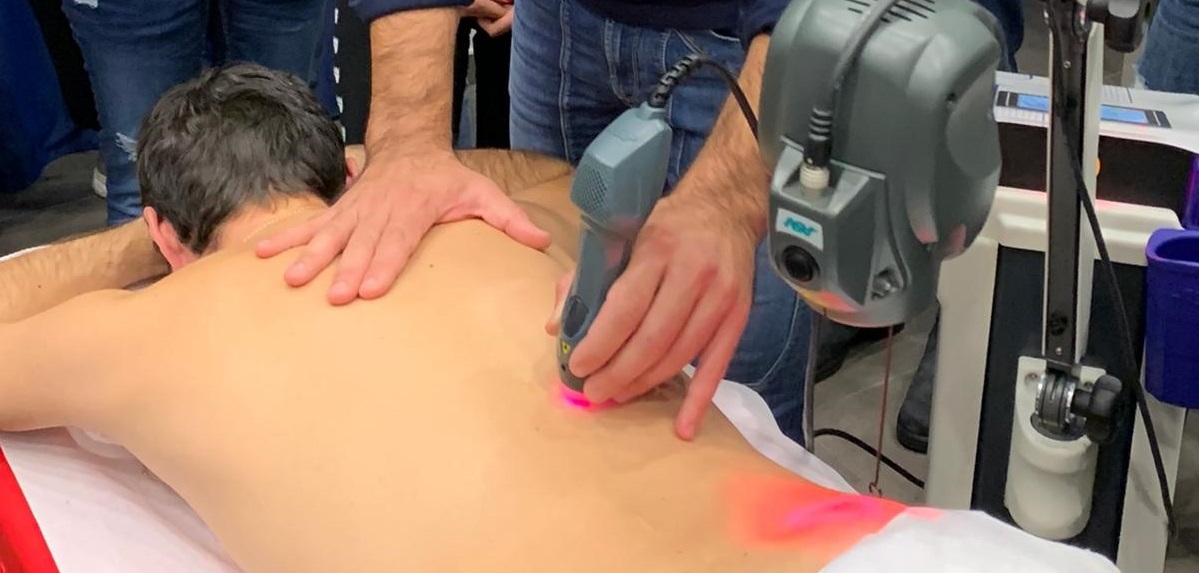
In the case of herniated discs, conservative treatments such as rest, manual manipulation or physical therapy techniques, including laser therapy, can alleviate the symptoms. If the patient does not respond to these treatments and/or the associated symptoms (i.e. weakness and numbness of the limbs affected by the nervous path) continue to deteriorate, he/she may undergo spinal surgery such as discectomy, spinal fusion or the application of an artificial intervertebral disk.
Laser therapy has proved to be a useful aid in the treatment of hernia pain, both at a cervical level (see clinical case: Dr Perez, Energy for Health # 14) and at the lumbar one (see clinical case: Dr Grennell, Energy for Health # 17).
One patient, already treated with painkillers and anti-inflammatories both locally and by mouth without results, was seen by Dr Grennell with a condition of VAS 10 (maximum pain possible). Treated with laser therapy for a few months, she showed tangible improvements in terms of pain and quality of life (sleep, mobility) right from the first sessions, so much so that she could undergo therapeutic exercise after a few weeks. The results of the treatment cycle showed constant and continuous progress, up to the complete removal of pain. A condition that the patient also confirmed at the follow-up exam 3 months after the end of the treatment. The case history presented by Dr Perez also confirms the efficacy of laser therapy in treating disc herniation: a patient showing persistent severe pain associated with cervical hernia, was able to completely eliminate the pain symptoms and avoid surgery thanks to laser therapy.






L'accesso alla visualizzazione dei prodotti e al materiale informativo è riservato agli operatori del settore in ottemperanza alla legislazione vigente. ASA richiede di qualificarsi come operatore del settore per procedere con la navigazione.
Decreto Legislativo 24 febbraio 1997, n°46 Articolo 21
1. E' vietata la pubblicità verso il pubblico dei dispositivi che, secondo disposizioni adottate con decreto del Ministro della Sanità, possono essere venduti soltanto su prescrizione medica o essere impiegati eventualmente con l'assistenza di un medico o di altro professionista sanitario.
2. La pubblicità presso il pubblico dei dispositivi diversi da quelli di cui al comma 1 è soggetta ad autorizzazione del Ministero della Sanità. Sulle domande di autorizzazione esprime parere la Commissione di esperti prevista dall'articolo 6, comma 3, del decreto legislativo
30 dicembre 1992, n. 541, che a tal fine è integrata da un rappresentante del Dipartimento del Ministero della Sanità competente in materia di dispositivi medici e da uno del Ministero dell'Industria, del commercio e dell'artigianato.
Some of the contents of this website cannot be disclosed in the USA and its territories and possesions, for regulatory reasons. If you are a US resident, please click on the button here below and access ASA's distributor website for North America.