





Building valuable relationships with its distributors, both local and international, is one of ASA’s priorities as it has always believed in the importance of setting up networking activities aimed at providing the opportunity for growth to all the parties involved.
An example of this was provided by the 2021 edition of TAPRM (Taiwan Academy of Physical and Rehabilitation Medicine) which was once again able to count on the active participation of Gaia Genomics, ASA’s partner in the Taiwan area, for this year’s annual meeting.


The meeting’s scientific schedule was led by Hilterapia® which was the focus of a lunch seminar held by Doctor Richard Malousek, Physician of the Austrian national Rowing Team and owner of a rehabilitation clinic in Vienna.
His speech - “Clinical experience of Hilterapia® on Sport injuries and spine diseases” – given at dawn from Vienna in order to be live in Taiwan – also showcased 4 interesting and documented clinical cases whose thorough analysis stimulated participants to further explore the therapy.

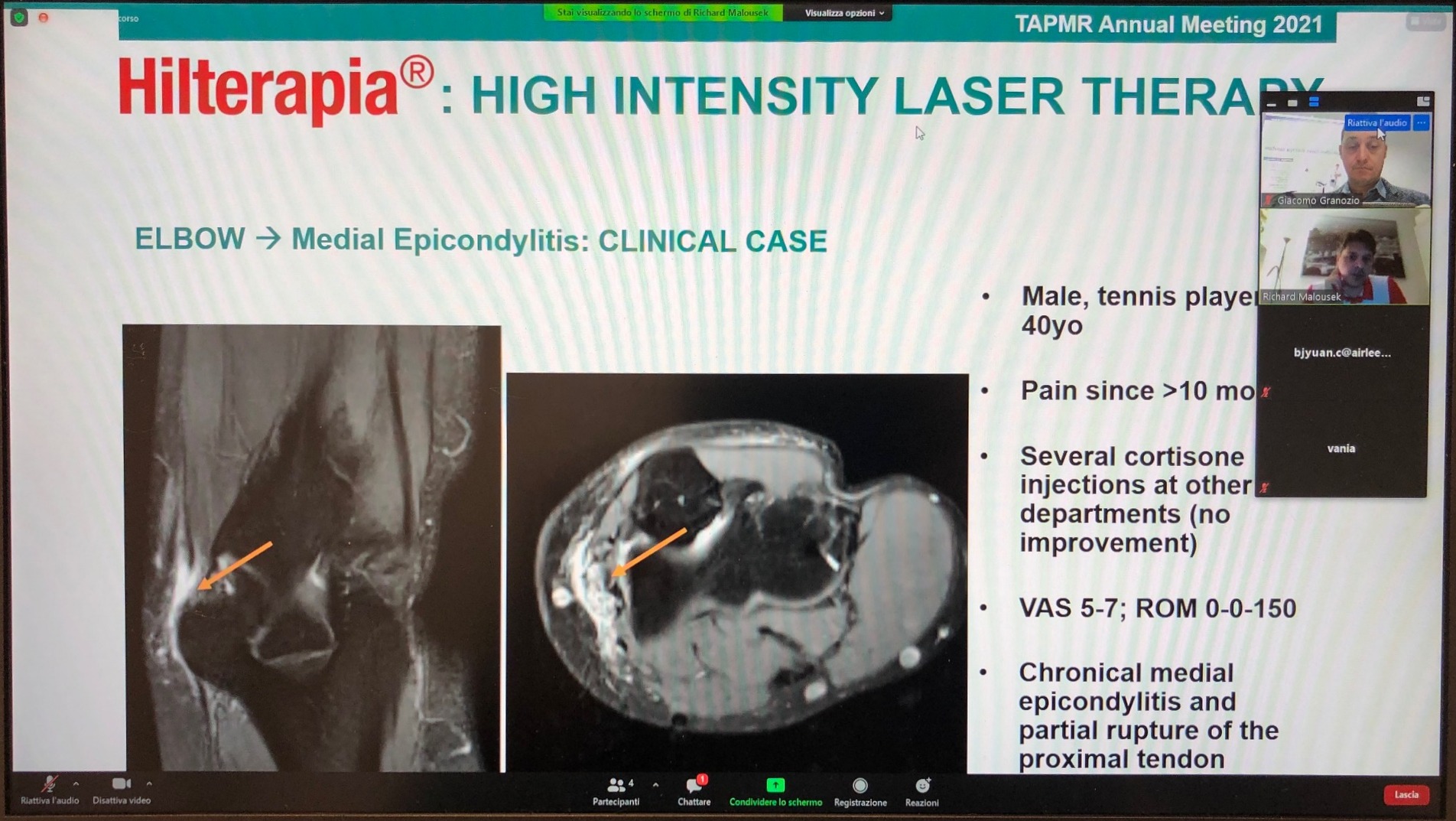
“An important result achieved on this occasion, which, in addition to allowing us to further strengthen our relationship with Gaia Genomics, highlighted the interest of industry professionals towards our therapy, thanks also to contributions by renowned specialists such as Doctor Malousek” Giacomo Granozio, Export Area Manager, explains.
Additional prestige to the event was also provided by the participation of Muyen Chu, gold medallist in Taekwondo at the Athens Olympics and the 2021 testimonial for Gaia Genomics, whose team took part in the meeting wearing an ASA kimono precisely in honour of the Olympic Champion.








L'accesso alla visualizzazione dei prodotti e al materiale informativo è riservato agli operatori del settore in ottemperanza alla legislazione vigente. ASA richiede di qualificarsi come operatore del settore per procedere con la navigazione.
Decreto Legislativo 24 febbraio 1997, n°46 Articolo 21
1. E' vietata la pubblicità verso il pubblico dei dispositivi che, secondo disposizioni adottate con decreto del Ministro della Sanità, possono essere venduti soltanto su prescrizione medica o essere impiegati eventualmente con l'assistenza di un medico o di altro professionista sanitario.
2. La pubblicità presso il pubblico dei dispositivi diversi da quelli di cui al comma 1 è soggetta ad autorizzazione del Ministero della Sanità. Sulle domande di autorizzazione esprime parere la Commissione di esperti prevista dall'articolo 6, comma 3, del decreto legislativo
30 dicembre 1992, n. 541, che a tal fine è integrata da un rappresentante del Dipartimento del Ministero della Sanità competente in materia di dispositivi medici e da uno del Ministero dell'Industria, del commercio e dell'artigianato.
Some of the contents of this website cannot be disclosed in the USA and its territories and possesions, for regulatory reasons. If you are a US resident, please click on the button here below and access ASA's distributor website for North America.